Reliance Agreements
A Reliance Agreement (also called an Institutional Authorization Agreement, or IAA) is an agreement signed by two or more institutions engaged in the same human subjects research project. The Agreement permits one or more institutions to cede review to another central, or single, IRB. Generally speaking, this means that only one IRB will review a single project for several collaborators across institutions. Select a tab in the menu to learn more about when and how to incorporate a Reliance Agreement to your project.
What Kind of Agreement Do I Need?
There are many ways to incorporate a non-USU researcher into your project, and not all of them are via a Reliance Agreement. This flowchart will help you determine which agreement is appropriate for your external colleague(s). All documents referenced for upload into Kuali are built into the Kuali Protocol template.
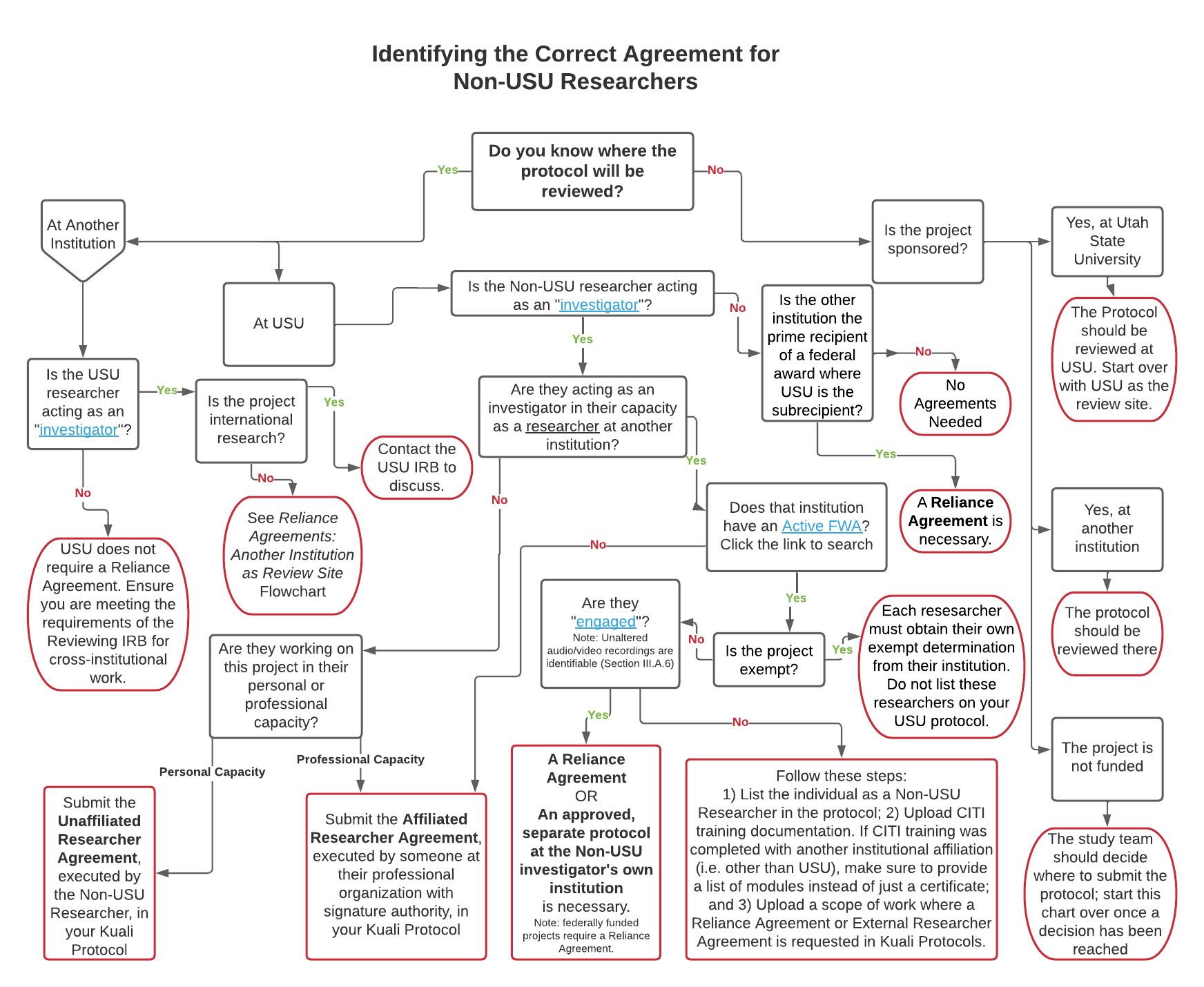
Identifying the Correct Agreement for Non-USU Researchers PDF
When to use a Reliance Agreement
A Reliance Agreement is needed if one of the following is true:
- You are a Principal Investigator (PI) on a human subjects research project reviewed at USU, and you plan to have non-USU investigators engaged in the project.
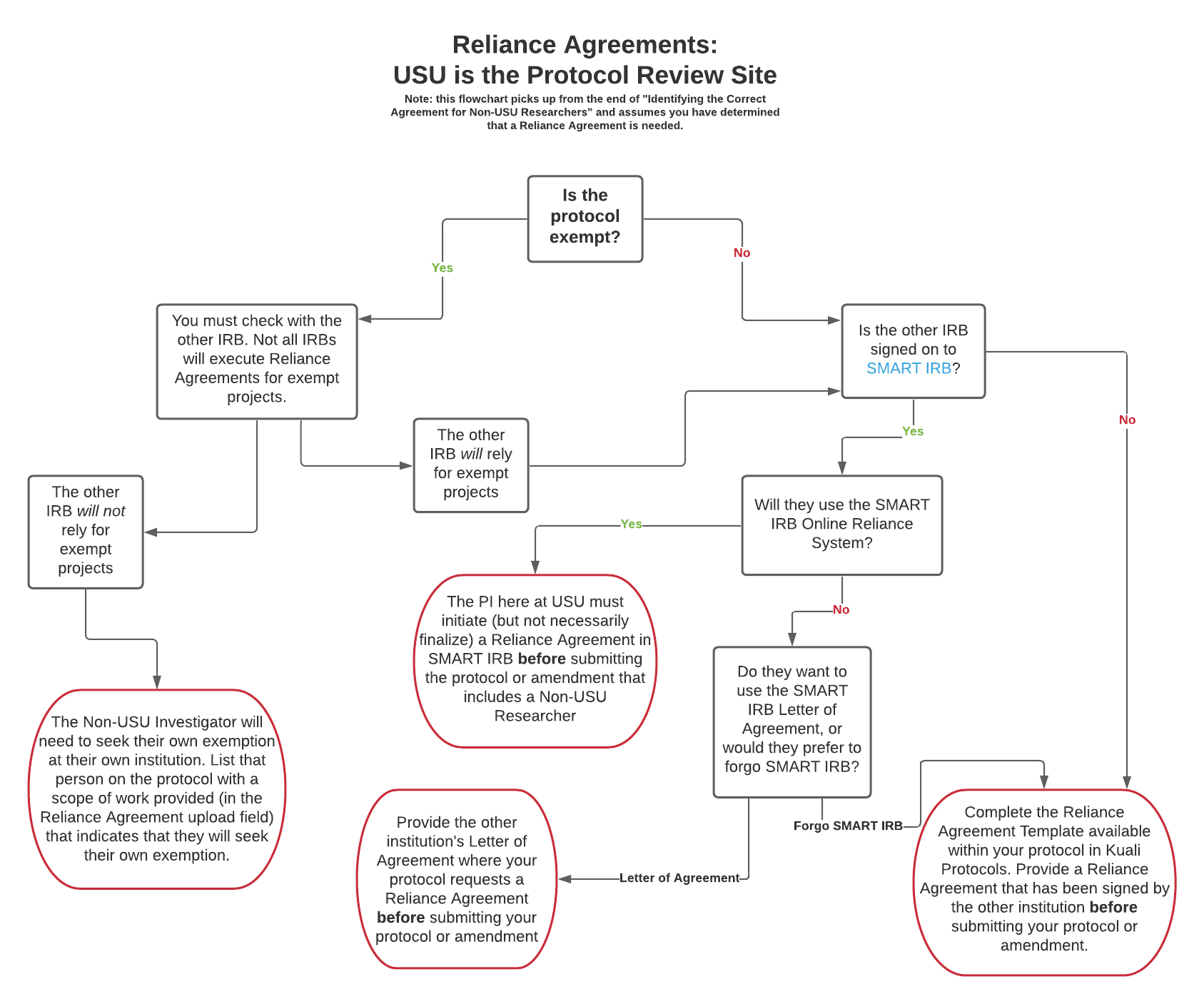
Reliance Agreements: USU is the Protocol Review Site PDF
2. You are USU researcher and you will be an investigator engaged in a human subjects research project, but the PI is at another institution. You will submit an External Reliance protocol in Kuali Protocols. This submission allows researchers to request that the USU IRB rely on another IRB's review and oversight of the project.
- In Kuali Protocols, your “type of review or action” will be “Reliance on Another IRB’s Review.” Once that is submitted by the USU local investigator, USU’s IRB will review the materials, make a determination about the eligibility for reliance, and send off appropriate requests for Reliance to the other IRB. You can view the status of your Reliance Request in the Admin Notes & Files section of your Reliance Request.
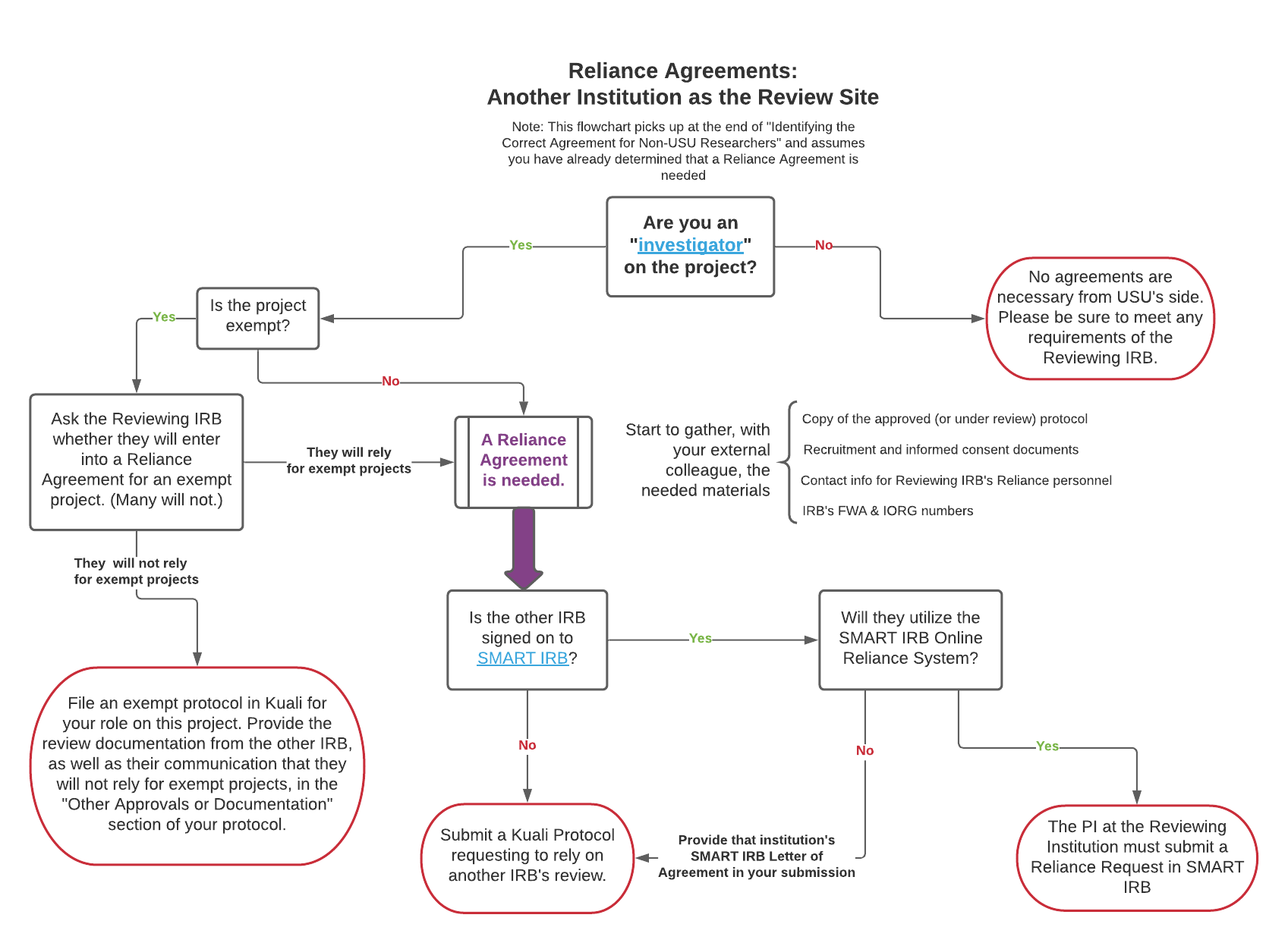 Reliance Agreements: Another Institution as the Review Site PDF
Reliance Agreements: Another Institution as the Review Site PDF
How to Request a Reliance Agreement
- If you are Requesting that the USU IRB rely on Another Institution’s IRB:
- Go to Kuali Protocols.
- Complete the first page of information. The Principal Investigator should be USU's Lead Investigator, even if that person is not the overall Principal Investigator for the collaborative project.
- At the end of that page, select that you are requesting Reliance on Another IRB's Review.
- Complete the remainder of the Reliance Request. You must provide:
- A copy of the approved or under review protocol at the Reviewing Institution.
- A copy of all recruitment & informed consent documents.
- An appropriate contact person at the other institution's IRB.
- If the Reviewing Institution has a Reliance Agreement, Institutional Authorization Agreement, or SMART IRB Letter of Agreement/Acknowledgement, it must be provided.
- Any other documentation the Reviewing IRB wants the USU IRB or USU Investigators to complete.
- If you are requesting that another institution’s IRB rely on the USU IRB:
- Check to see if the other institution uses SMART IRB. If they do, you will need to get them to execute USU's SMART IRB Letter of Agreement before submitting your protocol or amendment which includes a Non-USU Researcher.
- If the Reviewing IRB does NOT use SMART IRB, the other institution must complete a Reliance Agreement before the USU team submits a protocol or amendment which includes a Non-USU Researcher.
- If the other IRB requires that USU complete its review before they agree to rely, documentation to that effect from the other IRB must be provided where that researcher's Agreement is requested.
Reliance Agreement Considerations
Institutions vary as to whether and how they will utilize Reliance Agreements. Determinations regarding Reliance Agreements are highly specific to each institution and each protocol. For example, many IRBs (including USU's) will not enter into these kinds of agreements for Exempt projects. Other institutions may have created review types and standards that are specific to their human research protection program, and will not engage in Reliance activities for projects that utilize those review structures.
Here are some specifics regarding USU’s Reliance Agreement process which may be relevant as you engage with other IRBs about initiating reliance processes:
- Utah State University will not engage in Reliance for Exempt studies. Each investigator working on an Exempt study must have their own exemption in place at their own institutions.
- Utah State University will engage in Reliance for any institution holding a Federalwide Assurance with HHS. This includes international institutions.
- Utah State University will not engage in Reliance with institutions that do not hold a Federalwide Assurance. External researcher agreements or separate IRB oversight should be sought in those cases.
- Utah State University is a member of SMART IRB. However, Utah State University will not use the SMART IRB Online Reliance System (ORS). It uses a SMART IRB Letter of Agreement, which ensures that all relying institutions are abiding by the SMART IRB Master Agreement without having to make use of two separate systems.
- For institutions wanting information on specifics regarding USU, please see or share Utah State University's Institutional Profile.
- Utah State University cannot commit to entering into Reliance Agreements for emeritus faculty or other non-full-time Principal Investigators where there are other investigators who are actively employed by their Federalwide Assurance-holding institution. In such cases, protocol review should take place separately OR at the other institution, listing the emeritus faculty as an external researcher.
- Utah State University does not enter into Reliance Agreements on behalf of students. A faculty PI must be identified.
- USU’s Federalwide Assurance Number is FWA00003308 (expires 07/02/2029) and its IRB Registration, or IORG, number is 0000121.
