IBC/Biohazards Committee
Policy & Purpose
The existence of an Institutional Biosafety Committee (IBC) is required by the National Institutes of Health (NIH) for research involving recombinant and synthetic nucleic acids. In addition to recombinant DNA/nucleic acid, the Utah State University (USU) IBC reviews, approves and oversees project proposals involving biohazardous materials (including BSL-2/3 infectious agents), “Select Agents” (as stated in USU’s Policy 583.11: Safety and Health in Research), and dual use of research of concern (DURC) (required under the 2015 US Policy for Institutional Oversight of Life Sciences Dual Use Research of Concern).
The IBC shall coordinate its activities with the following USU institutions:
- Office of the Vice President for Research (VPR)
- Office of Research Integrity and Compliance (RIC)
- Office of Environmental Health and Safety (EH&S)
- Sponsored Programs Office (SPO)
- Institutional Review Board (IRB)
- Institutional Animal Care and Use Committee (IACUC)
- EHS Committees
- Chemical Hygiene Committee (CHC)
- Radiation Safety Committee (RSC)
- Risk Control Committee (RCC)
- University Safety Committee, and other oversight committees of the university, to ensure that research carried out at the USU meets the high ethical standards of the institution.
IBC meetings are generally open to the public. IBC meeting agenda and minutes are made available to the public upon request.
Charter & Standard Operating Procedures
IBC Charter Executed 07.26.18
PDF: IBC Standard Operating Procedures Revised 3.1.19
New Protocol Registration
Annual Reviews and Safety Reports (Adverse Events) coming soon.
Recombinant DNA/Nucleic Acid & Biohazardous Material
Definitions
In the context of the NIH Guidelines, recombinant and synthetic nucleic acids are defined as:
- Recombinant nucleic acid molecules that are constructed by joining nucleic acid molecules and that can replicate in a living cell,
- Synthetic nucleic acid molecules that are chemically, or by other means, synthesized or amplified and that may wholly or partially contain functional equivalents of nucleotides,
- molecules that result from the replication of those described in (i) or (ii) above.
Biohazardous materials are defined as materials of biological origin that have the capacity to produce deleterious effects on humans or animals. This includes:
- Infectious agents requiring handling conditions above Biosafety Level-1.
- Recombinant DNA/Nucleic Acid.
- Human and/or animal blood and blood products, body fluids, tissue, and/or cells.
- Biological toxins.
- Agents regulated by CDC or USDA (e.g. on the list of Select Agents).
* For more information on biohazardous materials, see the CDC’s Biosafety in Microbiological and Biomedical Laboratories.
Policy & Review Process
According to USU’s Policy 583.11: Safety and Health in Research any research proposals involving:
- The use of any hazardous materials which may expose Investigators or other persons to potentially unsafe conditions, including but not limited to Select Agents, and
- The use or manipulation of Recombinant DNA
Must be reviewed and approved by the proper oversight committee.
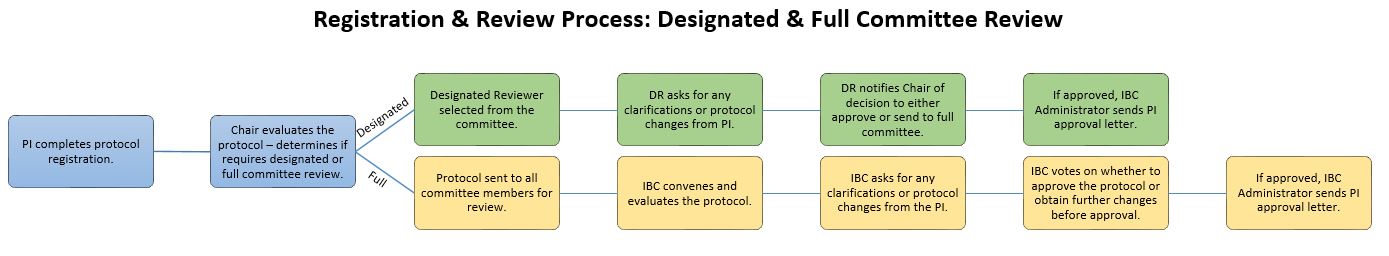
After PI completes protocol registration, the Chair evaluates the protocol and determines if it requires designated or full committee review.
If Designated:
- Designated Reviewer is selected from the committee
- DR asks for any clarifications or protocol changes from PI
- DR notifies Chair of decision to either approve or send to full committee
- If approved, IBC Administrator sends PI approval letter
If Full:
- Protocol is sent to all committee members for review
- IBC convenes and evaluates the protocol
- IBC asks for any clarifications or protocol changes from the PI
- IBC votes on whether to approve the protocol or obtain further changes before approval
- If approved, IBC Administrator sends PI approval letter
Dual Use Research of Concern
According to NIH, dual use research of concern is life sciences research that, based on current understanding, can be reasonably anticipated to provide knowledge, information, products, or technologies that could be directly misapplied to pose a significant threat with broad potential consequences to public health and safety, agricultural crops, and other plants, animals, the environment, material, or national security.
Research that involves any of the 15 select agents/toxins and/or uses one or more of the 7 experimental effects of concern listed below requires IBC approval.
Select Agents/Toxins List
- Avian influenza virus
- Bacillus anthracis
- Botulinum neurotoxin
- Burkholderia mallei
- Burkholderia pseudomallei
- Ebola virus
- Foot-and-mouth disease virus
- Francisella tularensis
- Marburg virus
- Reconstructed 1918 Influenza virus
- Rinderpest virus
- Toxin-producing strains of Clostridium botulinum
- Variola major virus
- Variola minor virus
- Yersinia pestis
Experimental Effects of Concern
- Enhances the harmful consequences of the agent or toxin.
- Disrupts immunity or effectiveness of an immunization against the agent or toxin, without clinical and/or agricultural justification.
- Confers to the agent or toxin resistance to clinically and/or agriculturally useful prophylactic or therapeutic interventions against that agent or toxin or facilitates their ability to evade detection methodologies.
- Increases the stability, transmissibility or the ability to disseminate the agent or toxin.
- Alters the host range or tropism of the agent or toxin.
- Enhances the susceptibility of a host population to the agent or toxin.
- Generates or reconstitutes an eradicated or extinct agent or toxin listed in the definition of DURC Agents.
Policy & Review Process
As per the US Government Oversight Policy on Dual Use Research of Concern, the risks and benefits of a project using DURC must be assessed and a Risk Mitigation Plan submitted, which is approved by IBC and US Government funding agency.
Process Steps:
- PI completes protocol registration
- Protocol sent to all committee members for review
- IBC convenes and evaluates the protocol
- IBC asks for any clarifications or protocol changes from the PI
- IBC votes on whether to approve the protocol or obtain further changes before approval
- If approved, the IBC sends the protocol to the USG funding agency for approval
- If approved, IBC Administrator sends PI approval letter
Any sharing of DURC knowledge, information, technologies, and/or products is subject to Export Control. As a PI of a DURC research project, if you have foreign nationals among your personnel, are traveling with and/or shipping your DURC research knowledge, information, technologies, and/or products, YOU MUST COMPLETE 1) Export Control Training and 2) Technology Control Plan (found under Export Control Forms on Export Control website). Export control violations can result in penalties and fines which may apply to an individual, the institution or both.
Training
According to NIH Guidelines, all IBC members, Principal Investigators, and laboratory staff must be trained in laboratory safety and biosafety.
Required Training IBC Members
- Institutional Biosafety Committee Member Training (via CITI)
- Dual Use Research of Concern
- Biosafety Course Overview
- Laboratory-Acquired Infections
- Biohazard Risk Assessment
- Medical Surveillance
- NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules
- Select Agents, Biosecurity and Bioterrorism (via CITI)
Research Specific Training for Principal Investigators
- For research with Recombinant DNA:
- NIH Recombinant DNA (rDNA) Guidelines (via CITI)
- For research with Select Agents/DURC:
- Select Agents, Biosecurity and Bioterrorism (via CITI)
- Dual Use Research of Concern (via CITI)
Training Available for IBC members, PI’s, & Personnel
- Biosafety/Biosecurity Training (via CITI)
- Laboratory Safety Training
- BSL II/III Training
- Animal BSL II/III Training
- Blood Borne Pathogen Training
Instructions for CITI Registration & Adding a Course
Registration
- Go to the CITI website.
- Click “Register” in the upper right corner of the page.
- Under “Select Your Organization Affiliation,” type Utah State University in the search box. Select Utah State University from the dropdown menu. Continue to step 2 on the CITI website.
- Enter your personal information. A secondary email address is optional but recommended. Continue to step 3 on the CITI website.
- Create your username and password. Select a security question and answer. Continue to step 4 on the CITI website.
- Enter your country of residence and select it from the dropdown menu. Continue to step 5.
- Indicate your interest in receiving Continuing Education Unit credit for completed CITI Program courses. Select an answer about participating in research surveys. These are voluntary programs but you must answer the questions to proceed. Continue to step 6 on the CITI website.
- Provide answers to the required questions (starred). Non-starred questions are optional. The institutional email address should be your email address affiliated with USU. If you do not have an office phone, please enter a mobile or home number. Continue to step 7 on the CITI website.
- Select the training you wish to complete and go to the next page.
- Click “Finalize Registration.”
Adding a Course
- Go to the CITI website and log in.
- Under “Main Menu/My Courses “scroll down to the bottom.
- Under “My Learner Tools for Utah State University”, click Add a Course.
- Select “Biosafety/Biosecurity courses” and click next.
- Select the course you wish to complete and click next.
- You should now be enrolled for that course.
IBC Responsibilities
The IBC holds meetings on a regular basis (usually monthly) to review new and updated protocols.
- In this review, the IBC assesses proposed project 1) containment levels, 2) facilities, 3) procedures, 4) practices, and 5) training and expertise of personnel
- The IBC may require changes to the protocol and approves the final version.
- The IBC Administrator notifies the PI of the results of their review via an approval letter.
- The IBC reviews meeting minutes; the Administrator maintains all records.
- The IBC continues to monitor all approved research protocols.
- The IBC reports any adverse events or compliance violations to the NIH Office of Science Policy.
In accordance with Section IV-B-2-a-(4) of the NIH Guidelines, IBC members must recuse themselves from review or approval of projects in which they hold a direct financial interest.
PI Responsibilities
The NIH Guidelines acknowledge that “[t]he safe conduct of experiments involving recombinant DNA depends on the individual conducting such activities.” For this reason, all Principal Investigators (PIs) shall become familiar with the NIH Guidelines, and shall adhere to their intent, as well as to the specific elements contained in them. In accordance with USU’s Policy 583.11, PIs shall obtain training available through the IBC, EH&S and other appropriate sources, and shall also ascertain that all laboratory personnel receive appropriate training to carry out their functions safely and in a scientifically appropriate manner. USU depends upon its PIs to use sound judgment when conducting research involving recombinant DNA/nucleic acids, biohazardous material, and DURC.
- PI’s must register their proposed protocol on ServiceNow.
- PI’s must address any IBC questions.
- PI’s must complete any required training.
- PI’s should NOT begin research until all required committees have given approval.
- PI’s must follow any conditions set forth by the IBC for their research protocol.
- PI’s must notify the IBC if any part of their research protocol (personnel, high risk activities, etc.) changes.
- PI’s must immediately complete a Safety Report Form if an adverse event occurs.
IBC Committee 2023-2024
The Institutional Biosafety Committee has combined with the Biohazards Committee in 2018.
The IBC is composed of no fewer than 5 voting members with the collective experience and expertise to assess the safety of research involving recombinant DNA/nucleic acid, biohazardous material, and DURC. The IBC may use consulting experts to execute its responsibilities or acquire needed expertise for select tasks. Consultants are not IBC voting members.
| Name | Member Type | Expertise | |
|---|---|---|---|
| Brian Gowen | Chair | Virology | brian.gowen@usu.edu |
| Ryan Jackson | Vice Chair | Recombinant DNA | ryan.jackson@usu.edu |
| Kirt Poulsen | Voting Member | Biological Safety Officer | kirt.poulsen@usu.edu |
| Ashley Dagley | Voting Member | Assistant Biosafety Officer | ashley.dagley@usu.edu |
| Aaron Olsen | Voting Member | Animal Lab Executive Director, Viral Pathology | aaron.olsen@usu.edu |
| Young-Min Lee | Voting Member | Virology | youngmin.ee@usu.edu |
| Diana Cox-Foster | Voting Member | Bacteriology | diana.cox-foster@ars.usda.gov |
| Charles Miller | Voting Member | Bio-Engineering | charles.miller@usu.edu |
| Sean Johnson | Voting Member | Biochemistry; RNA | sean.johnson@usu.edu |
| Nick Dickenson | Voting Member | Biochemistry, Bacteriology | nick.dickenson@usu.edu |
| Lee Rickords | Voting Member | Recombinant DNA | lee.rickords@usu.edu |
| Mike Weibel | Voting Community Member | Bear River Health Department | mweibel@brhd.org |
| Eric Jorgensen | Non-Voting Ex Officio | EHS | eric.jorgensen@usu.edu |
| Joy Van Nostrum | Non-Voting Ex Officio | Research Compliance | joy.vannostrand@usu.edu |